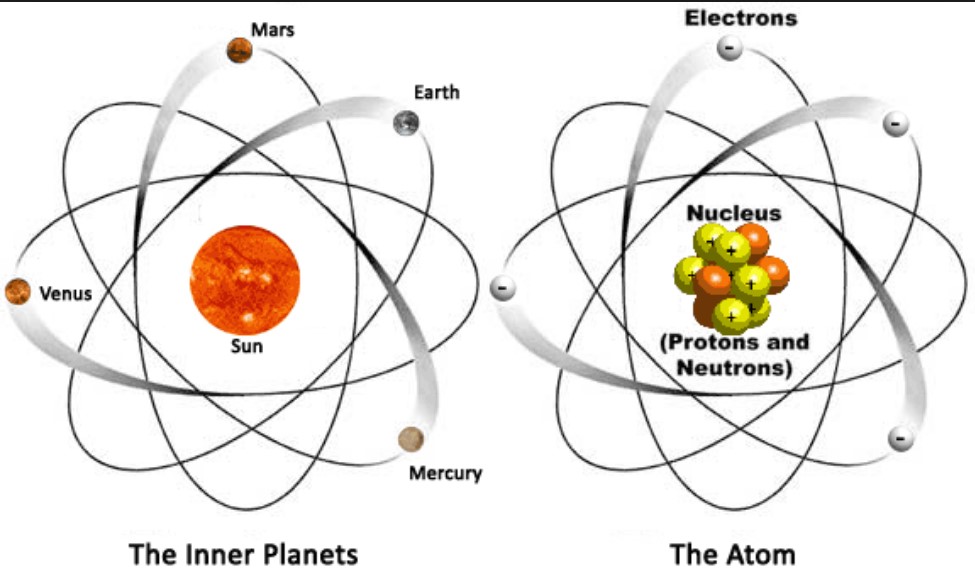
An atom consists of a cloud of electrons surrounding a small, dense nucleus of protons and neutrons. Atom Characteristics The number of protons in the nucleus is the atomic number of that atom. Each element is composed of one type of atom and can not be broken down into a simpler substance. Protons and electrons have electrical charges that are equal and opposite. Atoms are made of smaller particles, called electrons, protons, and neutrons. Atoms have a nucleus surrounded by an electron cloud Atoms are composed ofsmaller subatomicparticles in an atom.
Since the nucleus contains protons and neutrons, most of the mass of an atom is concentrated in its nucleus. The mass of an electron is very small compared to a proton or a neutron. In this second unit of class 11 chemistry, we will learn the. The relative mass of a proton is 1, and a particle with a relative mass smaller than 1 has less mass. They are the basis for everything in the Universe. When one says an atom is electrically neutral, it means that the number. The center of an atom is the nucleus and one or more electrons surrounding the nucleus. Instead of writing their actual masses in kilograms, we often use their relative masses. Most of an atom is just empty space and consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. As you learn more, you can move to the reactions and. Were going to cover basics like atomic structure and bonding between atoms. Neutrons are neutral, but protons and electrons are electrically charged. Solids are made of densely packed atoms while gases have atoms that are spread out. Answer (1 of 6): Atoms consist of electrons surrounding a nucleus that contains protons and neutrons. As you know, matter is composed of atoms. The masses of subatomic particles are very tiny. They are the basis for everything in the Universe. The structure of a carbon atom, not drawn to scale The nuclei of most atoms also contain neutrons. Utilizing radiation to combat cancer is an important career. The atom of the element Cobalt-60 is commonly used for this purpose. We would never have been able to treat cancer patients Gamma rays are used to kill off cancer cells and is used for cancer treatment. The nuclei of all atoms contain subatomic particles called protons. If the scientists did not explore the truth behind the atomic structure, 1.

The nucleus is tiny compared to the atom as a whole: The following is a simplified overview of the structure of the atom. An atom is the smallest constituent of a chemical element that still maintains the chemical properties of that element. This is surrounded by electrons arranged in shells. The structure of the atom is key to the majority of the techniques used in radiology, and a general understanding of atomic structure is worthwhile.
